We have previously developed a simulation-based proof-of-concept workflow to determine the mechanical fracture environment after tibial fractures in silico ( Braun et al., 2021). However, the translation of these mostly experimental or simulation-based parameters to real human patient clinical cases remains an absolute rarity. Strain quantities beneficial or at least not harmful for bone healing have been identified in simulations and experiments, such as, e.g., octahedral shear strain and hydrostatic strain ( Shefelbine et al., 2005), distortional strain ( Simon et al., 2011 Ren and Dailey, 2020) or deviatoric strain ( Son et al., 2014). 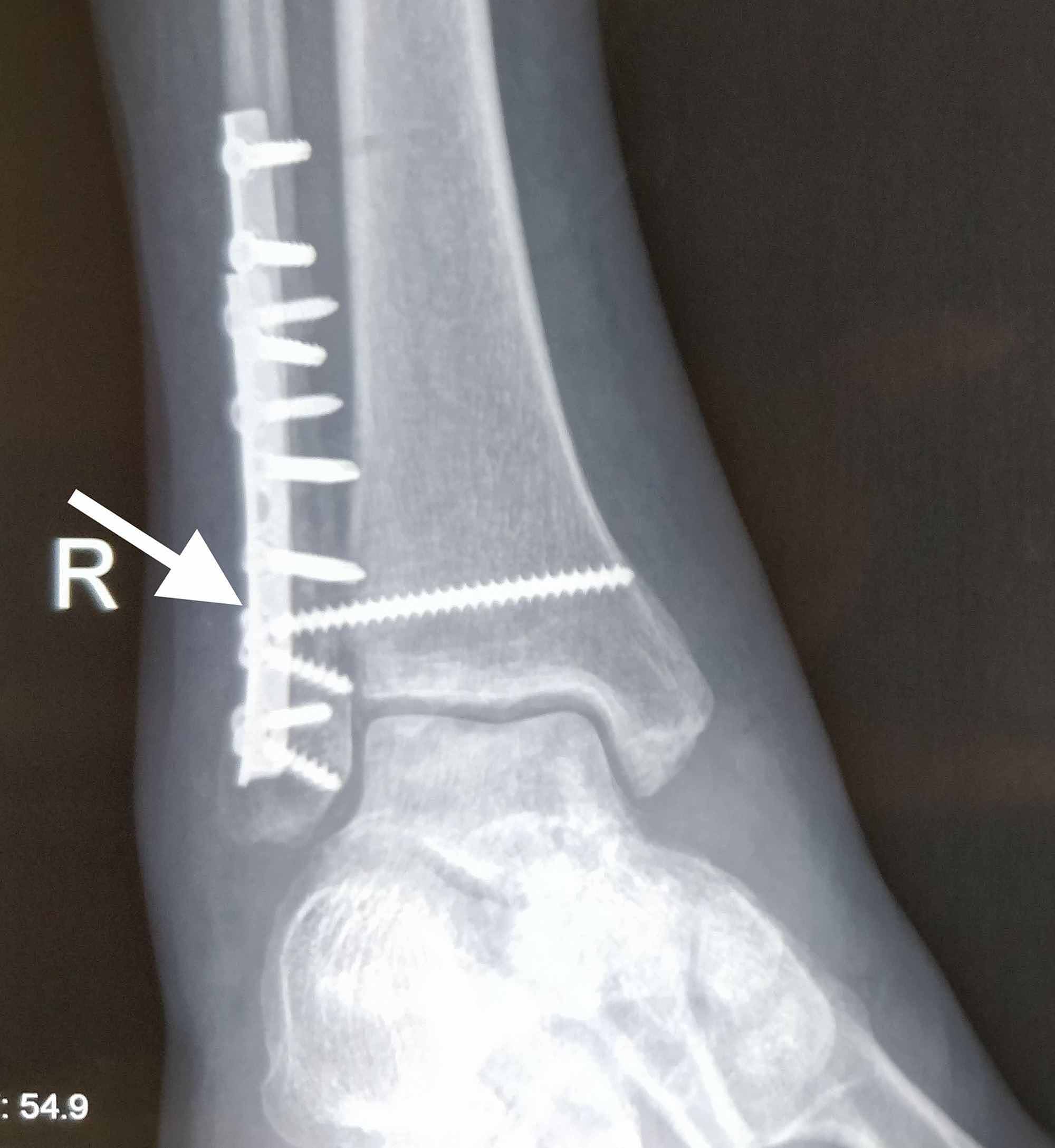
In fact, previous studies identified the mechanical parameters ‘interfragmentary strain’ and ‘hydrostatic pressure’, and their threshold values as reliable parameters to monitor the different types of bone healing such as, e.g., intramembranous and endochondral ossification ( Claes and Heigele, 1999 Shefelbine et al., 2005). Biomechanical modification, e.g., via implants may lead to a different clinical outcome ( Claes and Cunningham, 2009). Accordingly, the objective measurement of mechanobiological parameters helps to determine progress of the fracture healing process. During the postoperative course, they are often advised for restricted weight-bearing of the injured leg and are commonly followed up by regular clinical investigations and radiographic controls after at least after 6 and 12 weeks.Īlthough the etiology of disturbed bone healing may often be multifactorial, the mechanical environment within and around the fracture gap is known to be of crucial importance for the bone healing process ( Claes and Cunningham, 2009). To date, patients with fractures of the lower leg can be treated by a variety of surgical techniques. Simulations may improve decisions on the choice and location of surgical implants, as well as give recommendations for loading in the postoperative course of the individual patient.ĭespite recent experimental and clinical progress in the treatment of tibial and fibular fractures, the rates of delayed bone healing and non-union remain challengingly high ( Dailey et al., 2018). 
In conclusion, it is likely that motion, weight bearing and fibular mechanics influence the biomechanical milieu in the fracture gap. Weight-bearing restrictions were beneficial in reducing excessive mechanical conditions, while it is known that it is difficult for patients to adhere to partial weight-bearing recommendations. Moreover, the simulations showed that surgical treatment of the distal fibular fracture had an impact on the healing course, whereas the proximal fibular fracture barely mattered. In addition, a larger number of areas in the fracture gap with forces that exceeded beneficial mechanical properties longer was observed. The results show that a high postoperative walking speed was associated with higher loads in the fracture zone. The simulation of the real treatment was compared to the clinical course. The simulations were used to compute interfragmentary strain and the von Mises stress distribution of the intramedullary nail for different treatments of the fibula, as well as several walking velocities (1.0 km/h 1.5 km/h 2.0 km/h) and levels of weight-bearing restriction. Early postoperative motion data, recorded via an inertial measuring unit system and pressure insoles were recorded and processed to study strain. Based on the computed tomography (CT) data set of a real clinical case with a distal diaphyseal tibial fracture, a proximal and a distal fibular fracture, finite element simulations were run. The aim of this study was to simulate and compare different mechanical conditions after lower leg fractures to assess the effects of postoperative motion, weight-bearing restrictions and fibular mechanics on the strain distribution and the clinical course. 3Chair of Applied Mechanics, Saarland University, Saarbrücken, Germanyĭespite recent experimental and clinical progress in the treatment of tibial and fibular fractures, in clinical practice rates of delayed bone healing and non-union remain high.2Werner Siemens Endowed Chair of Innovative Implant Development (Fracture Healing), Saarland University, Saarbrücken, Germany.1Department of Trauma, Hand and Reconstructive Surgery, Saarland University, Saarbrücken, Germany.Marcel Orth 1* Bergita Ganse 1,2 Annchristin Andres 3 Kerstin Wickert 3 Elke Warmerdam 2 Max Müller 1 Stefan Diebels 3 Michael Roland 3 Tim Pohlemann 1 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
